Factorial clinical trial.

Factorial clinical trial allows testing more than one intervention with only one sample of participants.
The clinical trial is the king of epidemiological designs. But it is also the most expensive to perform. And, in our times, this is an important inconvenience to launch a trial.
Usually in each trial an intervention in one group versus the control group without intervention or placebo is evaluated. But, what if we could test various interventions in the same trial?. The costs probably would be lower than trying each intervention separately, each in its conventional parallel trial. Well, this is possible to carry out, using a design named as factorial trial.
Factorial clinical trial
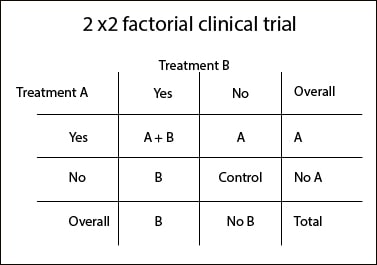
The simplest form is the 2×2 factorial trial, in which two different interventions are tried on the same sample of participants. The trick is to randomized participants several times to form more than the two groups of a parallel trial. Suppose we want to do a factorial trial with treatments A and B (let’s not complicate us too much thinking about an example).
First, we randomly assign participants to either receive or not the treatment A. Then we perform another randomization to receive or not treatment B. Thus, the sample of N participants is divided into four groups, as it’s shown in the attached table: N/4 receive only A, N/4 only receive B, N/4 receive A and B simultaneously, and N/4 remain untreated (control group).
This design is the basic 2×2 factorial trial. If we focus on the table, the analysis of marginal values in rows allows us to compare the effect between receiving A and not to receive it. For its part, the marginal analysis of the columns allows us to compare the effect between getting B and not to receive it. We could also compare values of each cell separately, but then we’d lose power to detect differences, and with that one of the advantages of this type of design.
The sample size required is usually calculated imagining that we are going to do two independent parallel trials and taking the largest number needed to detect the smallest of the effects we want to study.
Meanwhile, randomization is done using the same methods as with the parallel trial, but it’s repeated several times. Another alternative would be to identify all the groups (A, B, A + B and control, in our example) and make the random assignment at once. The result is the same.
The main reason for doing a factorial trial should be the economical one, since they require less participants than the two comparable parallel trials, so it results cheaper. This is especially useful if the sponsor of the trial does not expect to make huge profits with the results. Therefore it is common to see factorial trials with unprofitable, or with well-known and traditionally used treatments.
Conditions for conducting a factorial clinical trial
An important condition for making a factorial trial with guarantees is that there is no interaction between the two treatments, so that their effects are independent. When there is interaction between the two treatments (one effects depend on the presence of the other), the analysis is complicated and the necessary sample is higher, as we couldn’t analyze the marginal values of the table to detect differences, but we would have to assess the differences among all groups and, as we’ve said, the statistical power of the study would be lower.
In any case, we will always check for interaction. This can be done using a regression model with an interaction term and comparing the model with the same regression model without interaction. In the case of detecting interaction (which could not have been previously suspected), we must make an analysis of each group separately even at the cost of losing power to detect statistically significant differences.
And can we compare more than two interventions? We can compare all we want, but we must bear in mind that design complexity increases, as do the number of groups to be compared and the possibility of encountering interaction among any of the tested interventions. For these reasons, it is advisable to keep the number of interventions as low as possible.
We have already discussed the most obvious advantage of factorial trial: lower cost resulting of requiring a smaller sample size. Another advantage is that they serve if we are also interested in assessing the effect of the combination of interventions, assessing by the way the existence of interaction.
Meanwhile, interactions between interventions are the main limitation of this design. We have already mentioned that when there, we require the individual analysis of the groups, with the loss of power that entails. Another drawback is that the compliance of the participants cannot be very good: the more treatments a participant must perform correctly, the most likely he will not do it the way he should.
We’re leaving…
And here we leave for today the story of factorial clinical trials. We have described the simplest way, the 2×2 factorial. However, as we have said, things can get complicated comparing more interventions and also assigning different sizes to each of the groups. For example, if we detect smaller differences in one treatment group that interests us, we can assign more patients to it. Of course all this complicates the analysis and calculation of sample size. But that is another story…